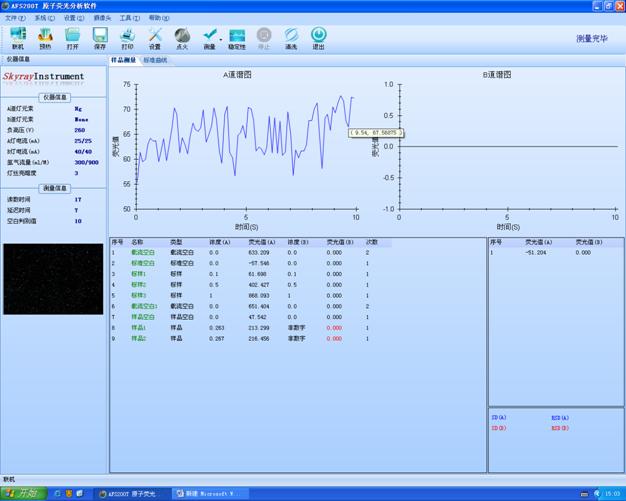
ЧА бд
НЈВФаавЕЪЧжаЙњживЊЕФжЪСЯЙЄвЕ�ЁЃНЈВФВњЦЗАќРЈНЈжўжЪСЯМАжЦЦЗЁЂЗЧН№ЪєПѓМАжЦЦЗЁЂЮоЛњЗЧН№ЪєаТжЪСЯШ§ДѓУХРр����ЃЌЙуЗКгІгУгкНЈжўЁЂОќЙЄЁЂЛЗБЃЁЂИпаТММЪѕЙЄвЕКЭШЫУёЩњЛюЕШСьгђ�ЁЃФПЧА����ЃЌжаЙњвбОЪЧЪРНчЩЯзюДѓЕФНЈжўжЪСЯЩњВњЙњКЭЯћЗбЙњ�ЁЃжївЊНЈВФВњЦЗЫЎФрЁЂЦНАхВЃСЇЁЂНЈжўЮРЩњжЪСЯЁЂЪЏВФКЭЧНЬхжЪСЯЕШВњСПЖрФъОгЪРНчЕквЛЮЛ�ЁЃЭЌЪБ����ЃЌНЈВФВњЦЗжЪСПВЛОјЬсИп����ЃЌФмдДКЭджЪСЯЯћКФж№ФъЯТНЕ����ЃЌжжжжаТаЭНЈВФВЛОјгПЯж����ЃЌНЈВФВњЦЗВЛОјЩ§МЖЛЛДњ�ЁЃдкПЦбЇММЪѕЗЩЫйЩњГЄЁЂИїЯюЪТвУїШеєеєШеЩЯШЗЕБНёЩчЛс����ЃЌЫцзХЮФЛЏКЭЮяжЪЩњЛюЫЎЦНЕФВЛЙмЬсИп����ЃЌШЫУЧдНЗЂзЂжиТЬЩЋНЁПЕЕФЯћЗбРэФю����ЃЌЖдЧщПідНЗЂжиЪг����ЃЌВЂЧвдНЗЂжиЪгТЬЩЋЛЗБЃ����ЃЌвђДЫЖдНЈВФВњЦЗЕФМьВташЧѓдНРДдНДѓ����ЃЌвВдНРДдНбЯИё����ЃЌетвЛЧаЖМРыВЛПЊжжжжМьВтвЧЦїУРЪЈЙѓБіЛсвЧЦїЙЋЫОЩњВњЕФжжжжОЋУмЦЪЮівЧЦї����ЃЌПЩвдТњзуНЈВФаавЕЕФжЪСППижЦашЧѓ����ЃЌЫѕЖЬЦЪЮіЪБМф����ЃЌВЂЧвЦЪЮіНсЙћзМШЗПЩПП����ЃЌДѓДѓМгЫйСЫЮвЙњНЈВФаавЕЕФЩњГЄ�ЁЃ
вЛЁЂдзгЮќЪеНЈВФаавЕНтОіМЦЛЎ
вЊСьНщЩмЃКБОвЊСьНщЩмСЫдзгЮќЪеЗЈВтЖЈНЈВФжаН№ЪєдЊЫиЕФОпЬхВтЪдвЊСь����ЃЌИУвЊСьжївЊеыЖдНЈВФжаН№ЪєдЊЫиЕФВтЖЈ����ЃЌОпгаПЩВйзїадКЭКмКУЕигІгУМлжЕ�ЁЃ
1ЁЂвЧЦї

2ЁЂбљЦЗДІРэ
НЈВФаавЕЕФжжРргааэЖр����ЃЌВюБ№ЕФбљЦЗЧАДІРэвЊСьвВВюБ№����ЃЌЦфжавЊСьАќРЈЫсШмЁЂМюШлЕШ�ЁЃ
3ЁЂБъзМХфжЦ
гУ1000ug/ml ЕФЕЅЮЛЫиБъзМжќДцШмвКж№МЖХфжЦГЩЫљашХЈЖШЕФЪТЧщШмвК�ЁЃ
4ЁЂЪЕМЪбљЦЗОйР§
НЈВФжаЭСПЕФВтЖЈ
бљЦЗДІРэЃКГЦШЁ0.5000gЪдбљжУгк100mLЩеБжа����ЃЌМг5mLбЮЫсЁЂ2ЁЋ3mLЯѕЫс����ЃЌжУгкЕчШШАхЩЯМгШШШмНт����ЃЌМгШы5mlИпТШЫс����ЃЌМгШШжСУАбЬ����ЃЌШЁЯТРфШД����ЃЌМгШы20mLЫЎ����ЃЌМгШШШмНтбЮРр�ЁЃРфШД����ЃЌвЦШы100mLШнСПЦПжа����ЃЌгУЫЎЯЁЪЭжСПЬЖШ����ЃЌЛьдШ�ЁЃЪдвКжаШчгаЬМЛЏЮяЁЂЙшЫсЕШГСЕэЮяЮіГі����ЃЌаыИЩЙ§ТЫ�ЁЃ
БъзМЧњЯпЕФЛцжЦЃКзМШЗвЦШЁ0mL����ЃЌ0.10mL����ЃЌ0.20mL����ЃЌ0.30mL����ЃЌ0.40mL����ЃЌ0.50mLЭБъзМШмвКжУгквЛзщШнСПЦПжа����ЃЌЛЎЗжМгШы2mLЯѕЫс����ЃЌгУШЅРызгЫЎЖЈШн����ЃЌдкгыеЩСПЪдвКЯрЭЌЬѕМўЯТ����ЃЌеЩСПЯЕСаБъзМШмвКЕФЮќЙтЖШ����ЃЌМѕШЅСуХЈЖШШмвКЕФЮќЙтЖШ����ЃЌвдЭЕФХЈЖШЮЊКсзјБъ����ЃЌЮќЙтЖШЮЊзнзјБъЛцжЦЪТЧщЧњЯп�ЁЃ
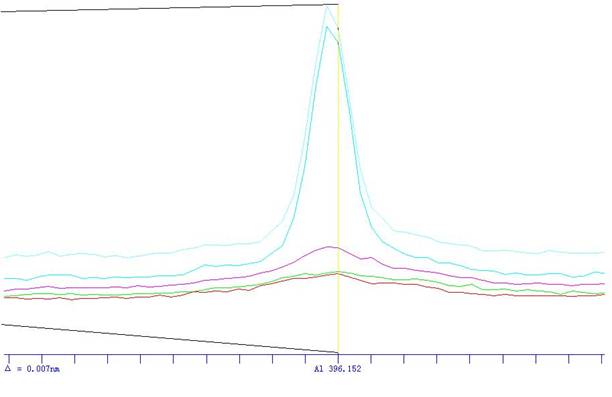
бљЦЗВтЪдЃКгкдзгЮќЪеЙтЦзвЧВЈГЄ324.8nmДІ����ЃЌЪЙгУПеЦј-ввШВЛ№бц����ЃЌвдЫЎЕїСу����ЃЌеЩСПЪдвКЕФЮќЙтЖШ����ЃЌМѕШЅЫцЭЌЪдСЯЕФПеАзЪдбщШмвКЕФЮќЙтЖШ����ЃЌДгЪТЧщЧњЯпЩЯВщГіЯргІЕФЭХЈЖШ�ЁЃ
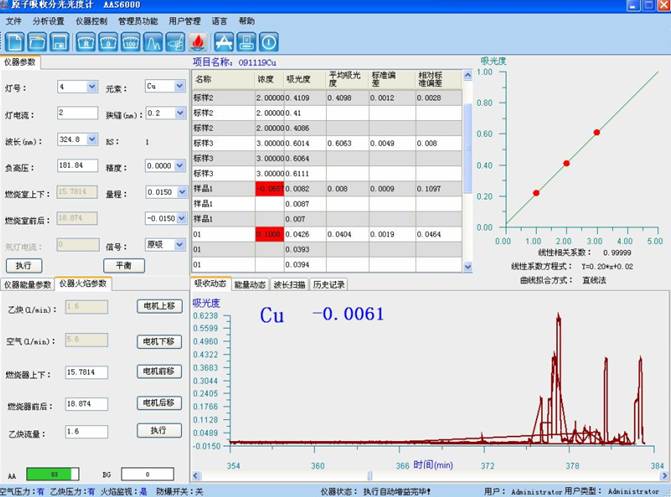
ЖўЁЂУРЪЈЙѓБіЛсвЧЦїICP-AESЗЈВтЖЈНЈВФжаН№ЪєдЊЫиНтОіМЦЛЎ
вЊСьНщЩмЃКБОвЊСьНщЩмСЫICPЃAESЗЈВтЖЈНЈВФжЪСЯжаН№ЪєдЊЫиЕФОпЬхВтЪдвЊСь����ЃЌИУвЊСьжївЊеыЖдКЯН№жаН№ЪєдЊЫиКЌСПЕФВтЖЈ����ЃЌОпгаПЩВйзїадКЭКмКУЕигІгУМлжЕ�ЁЃ
1. вЧЦї
ICPЃ2000аЭЕЅЕРЩЈУшЪНICPдзгЗЂЩфЙтЦзвЧ(УРЪЈЙѓБіЛсвЧЦї)

2. бљЦЗЬсШЁ
гЩПЭЛЇЬсЙЉОпгаДњБэадЕФбљЦЗ�ЁЃ
3. бљЦЗХфжЦ
ЦООнЪдбљжадгКЌСПЦщсЋНЋбљЦЗХфжЦГЩЪЪЕБХЈЖШ,ЫсЖШПижЦдк 5ЃЅзѓгвМДПЩ�ЁЃ
4. бљЦЗДІРэ
НЈВФжЪСЯЃЈВЃСЇЁЂЫЎФрЕШЃЉгааэЖр����ЃЌВюБ№ЕФбљЦЗЧАДІРэвЊСьвВВюБ№����ЃЌвЛАуЧАДІРэЫљгУЕФЪдМСгабЮЫсЁЂЧтЗњЫсЁЂЯѕЫсЁЂИпТШЫсЁЂЫЋбѕЫЎЕШ�ЁЃ
ЧАДІРэЃКГЦШЁ0.15gзѓгвбљЦЗгкВЃСЇЩеБЛђЪЧОлЫФЗњввЯЉЩеБжа����ЃЌЯШгУЯЁбЮЫсДІРэ����ЃЌД§ЗДгІЭъШЋ����ЃЌЦјХнМѕТ§Кѓ����ЃЌМгЃЈ1+1ЃЉЯѕЫсМЬајЯћНт����ЃЌжСЗДгІЭъШЋЃЈШчЙћНЈВФжаЙшКЌСПНЯИп����ЃЌОЭвЊЪЪЕБМгЧтЗњЫсДІРэ����ЃЌеєЗЂвдГ§ШЅЙшЃЉ����ЃЌЯћНтКѓЖЈШнжС100mLШнСПЦПжа,ЩЯЛњВтЪд�ЁЃЬиБ№ЪЧЫЎФр����ЃЌвЛАувЊгУЭѕЫЎЯћНт.
5. БъзМХфжЦЃК
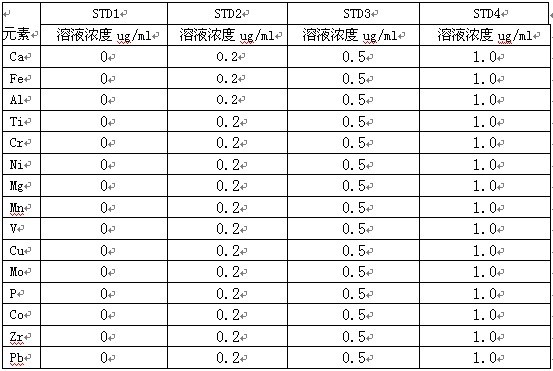
гУ1000ug/ml ЕФЕЅБъзМжќДцШмвКж№МЖХфжЦГЩ 100ug/mlШмвК����ЃЌдйж№МЖХфжЦГЩЛьЯ§БъзМШмвК10ІЬg/mlЁЂ1ІЬg/mlЫсЖШЮЊ10%(V/V)�ЁЃ
6. ICP-AESЪТЧщЧњЯпБъзМШмвКЕФХфжЦ
7. ЦЪЮіЯпЕФбЁдё
ЦООнICPШэМўЦзЯпПтЬсЙЉЫљгадЊЫиВюБ№ЦзЯп����ЃЌЪзЯШбЁдёБЛВтдЊЫиСщУєЖШИпЕФЦзЯп����ЃЌЦфДЮПМТЧВЛЪмбљЦЗЛљЬхМАБЛВтдЊЫиЯрЛЅжЎМфзЬШХаЁЕФЦзЯпЕФддђ����ЃЌбЁдёЦЪЮіЯпШчЯТ�ЁЃ
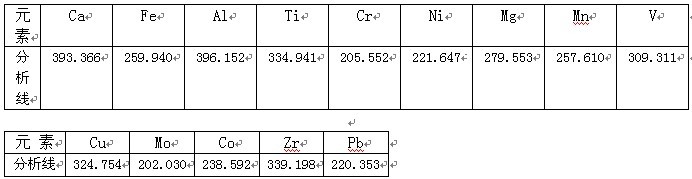
8ЃЎ НсЙћЪ§Он
вЊСьЭЈЙ§бЁдёзюМбЪТЧщЬѕМў����ЃЌНЈСЂСЫICPЃ2000аЭICPЃAESЗЈВтЖЈНЈВФжаН№ЪєдЊЫиВтЖЈвЊСь, ЕУГіЪЕбщЪ§Он�ЁЃЪЕбщжЄУїИУвЧЦїДѓВПЗжММЪѕжИБъЕжДяКЭНгНќСЫыЭтЭЌРрВњЦЗММЪѕжИБъ, гЩгквЧЦїВйзїШэМўЪЙгУжаЮФЯЕЭГ, ЪЧЙњШЫЪЙгУИќБуЕБ,ећИіЦЪЮіРњГЬПьЫйЁЂМђБуЁЂНсЙћзМШЗЁЂгаКмКУЕФИпаХМлБШЕШгХЕу, ЫљвдИУвЧЦївВЦеМАЕНИїИіЦЪЮіСьГЧ.
Ш§ЁЂAFS200TдкНЈВФаавЕгІгУЕФНтОіМЦЛЎ
вЊСьНщЩмЃКБОвЊСьНщЩмСЫдзггЋЙтЗЈВтЖЈНЈВФаавЕжаН№ЪєдЊЫиЕФОпЬхВтЪдвЊСь����ЃЌИУвЊСьжївЊеыЖдНЈВФжаН№ЪєдЊЫиКЌСПЕФВтЖЈ����ЃЌОпгаПЩВйзїадКЭКмКУЕигІгУМлжЕ
1ЁЂвЧЦїЃК

2ЁЂбљЦЗДІРэМАдЊЫиМьВт
2.1AsЕФВтЖЈ
2.1.1ЁЁAsЕФБъвКХфжУ�ЁЃ
ЪЙгУЩщ(As)БъзМЪЙгУШмвК(0.1ug/ml)����ЃЌгХМЖДПбЮЫсЃЈHCLЃЉ ����ЃЌЦЪЮіДПСђыхКЭПЙЛЕбЊЫс����ЃЌШЅРызгЫЎЃЈЕчзшТЪЁн10M ХЗФЗЃЉЕШЪдМС�ЁЃ

2.1.2диСївКЕФХфжЦ
дивКСїЭЈГЃбЁгУгыБъвКЛљЬхЯрвЛжТЕФЕШХЈЖШЫсШмвК����ЃЌгУгкЭЦЖЏбљЦЗжСЗДгІЯЕЭГМгШыЗДгІВЂЧхЯДећИівЧЦїЙмТЗ�ЁЃ
5%HCLЃЈЬхЛ§ЗжЪ§ЃЉ ЃКзМШЗСПШЁ 50ml ХЈбЮЫс����ЃЌгУШЅРызгЫЎЖЈШнжС 1000ml�ЁЃ
2.1.3ЛЙдМСЕФХфжЦ
0.5%ЧтбѕЛЏМиЃЈФЦЃЉ ЃЈK(Na)OHЃЉгы 2%Х№ЧтЛЏМиЃЈФЦЃЉ ЃЈKЃЈNaЃЉBH4ЃЉЛьЯ§ШмвК�ЁЃ
ХфжЦвЊСьШчЯТЃКЯШзМШЗГЦШЁ 5g ЧтбѕЛЏМиЃЈФЦЃЉШмгкШЅРызгЫЎжа����ЃЌЧыШЗЖЈЧтбѕЛЏМиЃЈФЦЃЉЭъШЋШмНтКѓ����ЃЌдйзМШЗГЦШЁ 20g Х№ЧтЛЏМиЃЈФЦЃЉЗХШыИУШмвКжа����ЃЌгУШЅРызгЫЎЖЈШнЕН 1000ml����ЃЌШмНтКѓвЁдШ�ЁЃНЈвщгУЪБЯжХф����ЃЌзюКУВЛвЊСєЫоЩњДц����ЃЌХфжЦСїГЬВЛПЩЕЙжУ�ЁЃ
2.1.4ЩщAs)ЕФбљЦЗДІРэ
ДгбљЦЗжаГЦСП0.20gзѓгвгкВЃСЇЩеБжа����ЃЌМгШы8ml HCl,5mlHF, 8ml HNO3�ЁЃМгШШЯћНт�ЁЃРфШДКѓЖЈШнгк100mlШнСПЦПжа����ЃЌМгШы5mlHCl����ЃЌ5%ЕФСђФђКЭПЙЛЕбЊЫсШмвК20ml�ЁЃЃЈЭЌЪБзібљЦЗПеАзЃЉ�ЁЃ
2.1.5вЧЦїВйзїЬѕМўЃК
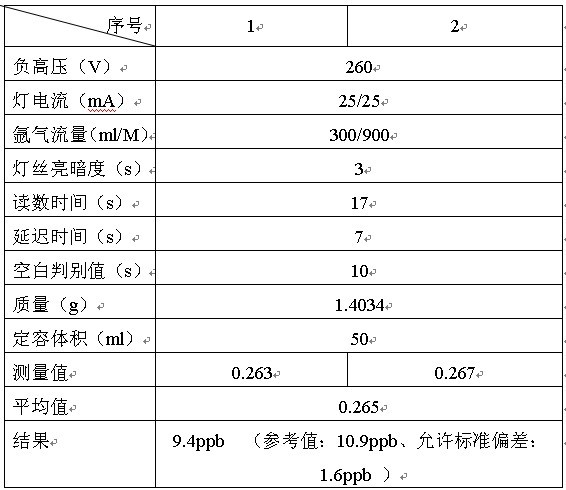
ИКИпбЙЃЈVЃЉ:270�ЃЛЕЦЕчСїЃЈmAЃЉ:40/40�ЃЛыВЦјСїСПЃЈml/MЃЉ:300/800
ЕЦЫПССАЕЖШ:3 �ЃЛЖСЪ§ЪБМф:12s�ЃЛбгГйЪБМф:2s�ЃЛПеАзХаБ№жЕ:2
3ЁЂгІгУЪЕР§
3.1.ЭПСЯжаВтЙЏ
ЧАДІРэЃКДгбљЦЗжаГЦСП1.4034gгкВЃСЇЩеБжа����ЃЌМгШы8ml HNO3КЭ2mlHClO4МгШШШмНтЃЈЯћНтЮТЖШ350ЁцзѓгвЃЉ�ЁЃРфШДКѓЖЈШнгк50mlШнСПЦПжа����ЃЌМгШы1ml HNO3����ЃЌ5%ЕФжиИѕЫсМи1ml�ЁЃЃЈЭЌЪБзібљЦЗПеАзЃЉЃЈЛЙдМСМг0.5%ЬњЧшЛЏМиЃЉ
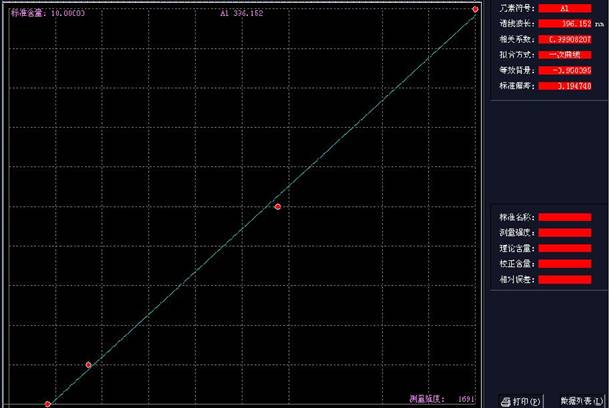
БъзМШмвКХфжУХЈЖШЃК0.1ppb 0.5ppb 1ppbзїЧњЯп�ЁЃ
ЃЈAЕРЯпадЗНГЬЃКY=875.144*X-17.003ЃЉ�ЃЛЃЈAЕРЯпаджЕЃК0.99916ЃЉМћЭМ1
вЧЦїВйзїЬѕМўЃК
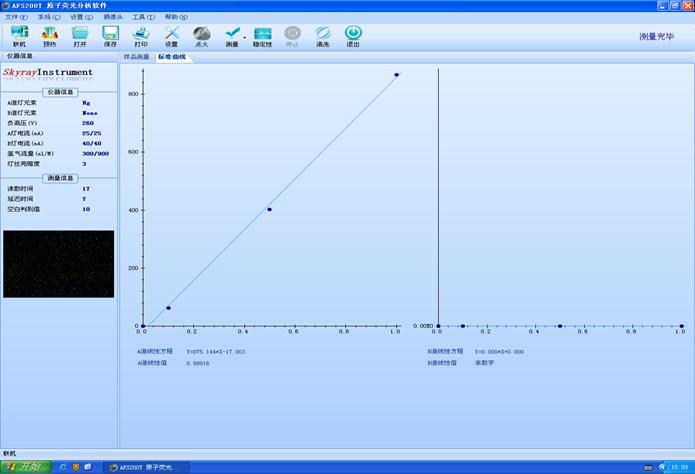
ЭМ1